Abstract
Introduction: Allogeneic hematopoietic stem cell transplantation (ASCT) remains a curative treatment for patients with advanced lymphomas. Haploidentical donors (HID) have recently extended transplantation for patients without HLA matched donors. Fludarabine (F) with melphalan (M) 140mg/m2 is the current standard of care conditioning regimen for ASCT. We hypothesized that progression-free survival (PFS) of patients with HID is comparable to that of HLA matched transplants, and that lower doses of melphalan (100mg/m2) are equally effective for these patients. Here, we analyzed outcomes of all lymphoma patients who received ASCT from different donors with FM conditioning at our institution between 09/2009-01/2018.
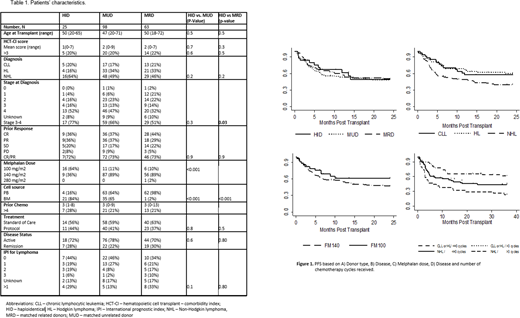
Methods: A total of 186 pts (63 matched related, MRD, 98 matched unrelated, MUD and 25 HID) were included. 30 patients had chronic lymphocytic leukemia (CLL), 54 Hodgkin lymphoma (HL) and 77 Non-Hodgkin lymphoma (NHL). The median follow-up was 57 (range 3-101) months. Patients received F160mg/m2 with M140mg/m2 (FM140) or 100mg/m2 (FM100). FM100 regimen was used in older patients/significant comorbidities. Thiotepa 5mg/kg or TBI 200cGy was added to HID transplants. All matched transplants received standard graft-versus-host disease (GVHD) prophylaxis (tacrolimus + methotrexate +/-ATG for MUDs), while HID group received PTCy-based GVHD prophylaxis. The cumulative incidence (CI) of non-relapse mortality (NRM), disease progression, and GVHD was estimated accounting for competing events. Regression analysis accounting for competing events was used to evaluate predictors of outcomes.
Results: Patients' characteristics were well balanced in donor groups except for advanced stage 3-4 disease (HID 77% vs MRD 51%, p=0.03), use of FM100 regimen (HID 64% vs. MUD 11% vs. MRD 10%, p<0.001) and bone marrow graft (HID 84% vs. MUD 36% vs. MRD 2%, p<0.001) (Table 1). Engraftment was similar in HID, MUD and MRD (92%, 89% and 98%, respectively; p=0.7). The CI of grade III-IV aGVHD at 6-months in HID (reference), MUD and MRD was 4%, 14% (p=0.2) and 8% (p=0.5), and the 3-year cGVHD were 5%, 16% (p=0.2) and 26% (0.09), respectively. The 3-year relapse for the HID (reference), MUD and MRD groups was 15%, 21% (p=0.4) and 39% (p=0.04), respectively. The corresponding CI of NRM was 31%, 32% (p=0.9), and 10% (p=0.02). The 3-year PFS was equivalent in the 3 donor groups at 49% (HID, reference), 44% (MUD, HR=1.2, p=0.6), and 46% (MRD, HR=1.1, p=0.8). Similarly, overall survival (OS) did not differ with respective OS of 52% (HID, reference), 54% (MUD, HR=1.04, p=0.9), and 67% (MRD, HR=0.6, p=0.2) in the 3 groups. The 3-year GVHD-relapse free survival in HID (reference), MUD and MRD groups was 39%, 31% (p=0.3) and 24% (p=0.2) respectively. Patients receiving FM100 were older (median 57 vs. 46 years, p<0.01), more likely to receive HID transplant (48% vs 6%, p<0.01) and a bone marrow graft (55% vs 25%, p=0.01), and less likely (12% vs 35%, p=0.01) to be treated for HL. On univariate analysis, FM100 regimen showed a trend for improved PFS (HR=0.7, p=0.3) and less relapse (HR=0.4, p=0.1) compared to FM140 regimen with similar NRM (HR=0.9, p=0.8) and OS (HR=0.9, p=0.7). The 3-year PFS and OS for FM100 vs. FM140 was 57% vs 43%, and 64% vs 58%. In multivariate analysis (MV), factors associated with unfavorable PFS were NHL (HR=1.9, p=0.002) and >3 chemotherapy regimens (HR=1.8, p=0.01) received prior to transplant. Including donor type and melphalan dose in MV analysis did not alter these results.
Conclusions: HID transplants have lower relapse rates and higher NRM comparing with MRDs, while no significant differences were seen comparing with MUDs, and similar survival compared with HLA matched transplants in patients with lymphoma receiving FM-based conditioning regimen. Our results suggest that FM100-based conditioning, which was used primarily in older individuals, is equivalent to FM140 and could be used as prefered standard conditioning regimen for ASCT for patients with lymphoma. Controlled studies are need to compare FM100 with FM140 conditioning and to evaluate PTCY-based GVHD prophylaxis in HLA matched transplants.
Oran:ASTEX: Research Funding; AROG pharmaceuticals: Research Funding; Celgene: Consultancy, Research Funding. Shpall:Affirmed GmbH: Research Funding. Fowler:Pharmacyclics: Consultancy, Research Funding; Janssen: Consultancy, Research Funding. Westin:Novartis Pharmaceuticals Corporation: Membership on an entity's Board of Directors or advisory committees; Apotex: Membership on an entity's Board of Directors or advisory committees; Kite Pharma: Membership on an entity's Board of Directors or advisory committees; Celgen: Membership on an entity's Board of Directors or advisory committees. Champlin:Otsuka: Research Funding; Sanofi: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal